Although PVC can be produced from a range of hydrocarbons including coal, and derivatives of plants such as sugar cane, the bulk of world production is currently manufactured using the chemical compound ethylene.
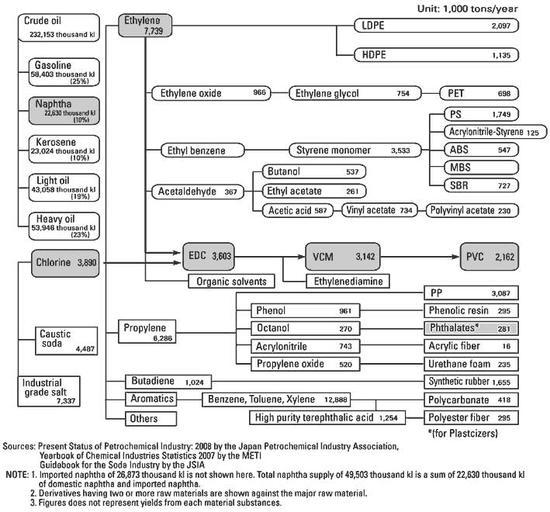
Ethylene (or ethene) which has the chemical formula C2H4, is a product of the oil & gas industry. PVC is therefore considered to be a petrochemical product. At the start of the ethylene-based PVC production process, the ethylene is combined with chlorine to produce an intermediate chemical known as EDC (ethylene dichloride or 1,2-dichloroethane).
Almost all EDC is used for PVC production in Europe, although a small portion is used for manufacturing of ethylenediamine, organic solvents and various pharmaceutical products.
Three major applications, polyethylene (PE), EDC and styrene monomer (SM) account for about 78% of all ethylene consumption (almost all styrene is used for PS).
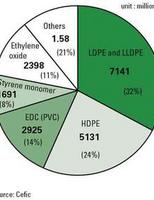
The petroleum refining industry and the basic petrochemical industry in Western Europe are predominantly located in coastal areas or where rivers or pipelines provide easy access to imported natural resources such as crude oil. They form petrochemical complexes, where refineries, crackers and petrochemical plants are connected by pipelines.
The chlor-alkali (caustic soda) industry is often located together with petrochemical complexes or near deposits or rock salt. VCM plants, which use ethylene and chlorine as major raw materials, and PVC plants, are generally located in petrochemical complexes due to this background.
Ethylene which is one of five types of products that are yielded by cracking of naphtha, and downstream on to the production of petrochemical products such as general purpose plastics.
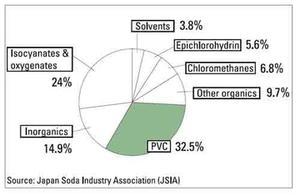
PVC and chlorine
Ethylene and chlorine are the major raw materials for VCM. Therefore, VCM is affected by the supply-demand conditions of both ethylene and chlorine, respectively. In Western Europe the share of VCM amounts to 14% of all ethylene use (ethylene requirement).
In contrast, VCM amounts to 35% of all chlorine use. This is why the demandsupply balance of chlorine has a greater impact on VCM than that of ethylene.
The balance between chlorine and caustic soda
Chlorine is a by-product of caustic soda production, generated at a ratio of 0.88:1. Applications for chlorine and caustic soda are totally different.
Therefore even if the balance between supply and demand for one of them is in equilibrium it does not necessarily follow that supply and demand for the other would be also.